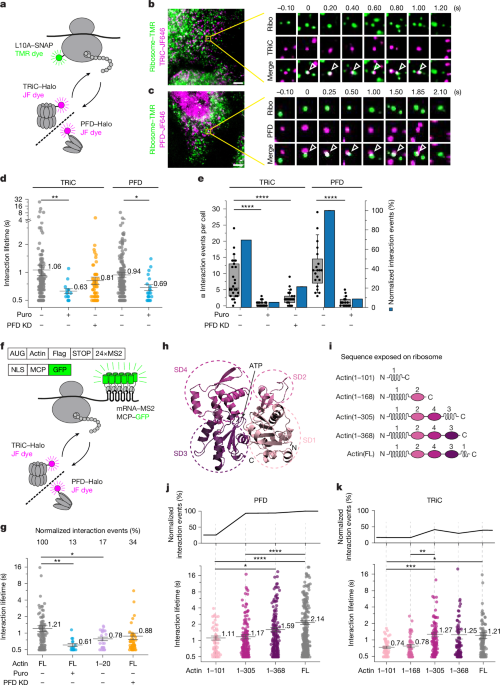
The essential chaperonin T-complex protein ring complex (TRiC) (also known as chaperonin containing TCP-1 (CCT)) mediates protein folding in cooperation with the co-chaperone prefoldin (PFD)1–5. In vitro experiments have shown that the cylindrical TRiC complex facilitates folding through ATP-regulated client protein encapsulation6–9. However, the functional dynamics of the chaperonin system in vivo remain unexplored. Here we developed single-particle tracking in human cells to monitor the interactions of TRiC–PFD with newly synthesized proteins. Both chaperones engaged nascent polypeptides repeatedly in brief probing events typically lasting around one second, with PFD recruiting TRiC. As shown with the chaperonin client actin8, the co-translational interactions of PFD and TRiC increased in frequency and lifetime during chain elongation. Close to translation termination, PFD bound for several seconds, facilitating TRiC recruitment for post-translational folding involving multiple reaction cycles of around 2.5 s. Notably, the lifetimes of TRiC interactions with a folding-defective actin mutant were markedly prolonged, indicating that client conformational properties modulate TRiC function. Mutant actin continued cycling on TRiC until it was targeted for degradation. TRiC often remained confined near its client protein between successive binding cycles, suggesting that the chaperonin machinery operates within a localized ‘protective zone’ in which free diffusion is restricted. Together, these findings offer detailed insight into the single-molecule dynamics and supramolecular organization of the chaperonin system in the cellular environment. Single-particle tracking experiments in intact cells reveal dynamic co- and post-translational interactions of the TRiC–PFD chaperonin complex with client proteins during in vivo protein folding.
Plasmids
To visualize co-translational interactions, plasmids encoding human β-actin, a 3×Flag tag, and 24 MS2 stem-loop repeats were constructed. The β-actin-24×MS2 (MBSV5) sequence54 (Addgene #102718) was amplified and subcloned into pRetroQ-AcGFP-... [26552 chars]
